| |
|
 |
The Evolution
of Chronic Kidney Disease: Defining a Model for Care World Congress of Nephrology (ASN/ISN), San Francisco, CA, October, 2001 Optimizing Anemia Management in Patients with CKD |  |
|
00:00

Optimizing Anemia Management in Patients with CKD
AJAY SINGH, MD: Thank you Bill. We're going to move along now to our next speaker. This is Dr. Robert Provenzano. He is the chair of the Department of Nephrology at St. John's Hospital and Medical Center in Detroit, USA. Bob has been interested in the area of anemia management, particularly pertaining to patients with chronic kidney disease for many years. The title of his talk is "Optimizing Anemia Management in Patients with Chronic Kidney Disease."
DR. ROBERT PROVENZANO: Thanks Ajay. Good evening. I hope everybody is relaxed. I appreciate you making time tonight in your busy schedules to listen to this important information.
00:00

Anemia Therapy: Overview
What I would like to do today is to touch on anemia therapy. This is a brief overview. As has already been pointed out, anemia management is less than optimal in our patient population with CKD. I'm going to talk a little bit about screening, about treatment options, both with epoetin alfa and darbepoetin, which has recently been approved for management in this patient population.
I'm going to spend a little bit of time speaking on iron supplementation, which is a very important aspect of therapy in this anemia. Then I'll spend a few minutes, as time allows, on optimal dosing of epoetin alfa.
00:00
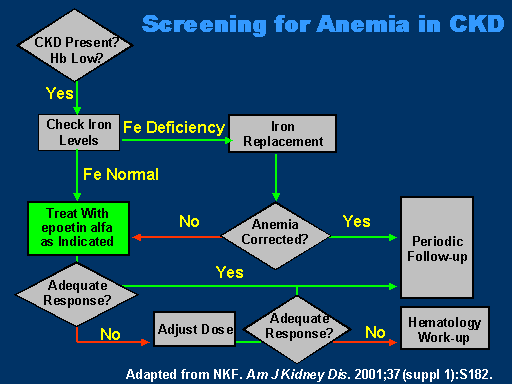
Screening for Anemia in CKD
This is a schematic. I know it's a little bit busy, but most of it is self-evident. The National Kidney Foundation Clinical Practice Guidelines, the NKF-K/DOQI guidelines, recommend that when CKD is present, that anemia be checked for. Short of getting into the details, one of the most important aspects before therapy is initiated, is checking iron levels.
Just following through the schematic, if iron deficiency is present, iron supplementation should be initiated. If, indeed, the anemia is corrected, periodic follow-up and monitoring is the next step.
If your iron levels are normal or after proper iron replacement anemia is still present, treatment with epoetin alfa is indicated. Based on the response, periodic follow-up, once you hit the target range, is done. Or if, indeed, with inadequate response, a hematologic workup, up to and including bone marrow, is the next step. What I'm going to do is touch on some aspects of this schematic to bring us to some greater detail.
00:00

Anemia Management: Initiation of Therapy
There are two aspects of anemia management. First is the initiation of therapy and then the follow-up therapy. Initiation of therapy after anemia is diagnosed involves first having a target. As mentioned, NKF-K/DOQI guidelines recommend a hemoglobin target of 11-12 grams per deciliter. As I mentioned, supplementation of iron is necessary, either prior to initiation of epoetin alfa therapy or simultaneously.
As I mentioned, there are now two medications available, both darbepoetin alfa and epoetin alfa. Then monitoring the hemoglobin and adjusting doses as necessary to reach this target.
Just briefly on these two medications -- and I will digress for just a moment. Although we all have our methods of dosing epoetin alfa in these patients, the logistics often become very difficult. I would suggest to you that although you may have your own protocol, you should certainly look in your clinic as to what is the best method. You'll find that this weighs heavily on physician time. Protocols should be set up in your practice so that either your medical assistant or your nurse practitioner or physician's assistant can deal with and understand the methods for dosing the particular medication which you choose to use.
00:00

Anemia Management: Maintenance
But very briefly, for epoetin alfa, increasing the dose by 50% if your hematocrit doesn't increase by at least 2% over 2-4 weeks and/or dropping the dose by 25% in a rapid responder if your hematocrit increase is greater than 8% in a month.
The recommendations for darbepoetin also. If your hemoglobin increase is less than 1 gram over 4 weeks and your iron stores are adequate, you would increase the dose 25%. Or if your hemoglobin increase in a rapid responder is more than 1 gram in a 2-week period, you would subsequently drop it by 25%.
00:00

Iron Deficiency
I want to spend a couple of minutes on iron deficiency. This is something that is often overlooked. There is some controversy about the best methods for replacing iron. Iron deficiency is still one of the more common reasons for resistant anemia. These patients tend to have accelerated -- tend to? They have certainly accelerated erythropoiesis, and you may underestimate their iron requirements. So monitoring their iron levels becomes much more important than in a non-CKD population.
Additionally, as will be pointed out later, therapy needs to be made cost effective. There are much data out there now that the cost effectiveness is markedly improved when iron is replaced. Indeed, even in iron-replete patients, there are some cost benefits to continue an iron supplementation.
00:00

Assessment of Iron Status
The typical assessments that we use include serum ferritins and transferrin saturation, maintaining your ferritin levels greater than 100 and less than 600 and your saturations greater than 20%.
There has been some argument that this really doesn't measure the available iron. There are some newer measurements out there that are becoming more clinically available. Indeed, I think there were a couple of posters in the last two days on reticulocyte hemoglobin content, erythrocyte ferritin and percent of hypochromic RBCs that may give us a better idea of the actual availability of iron in this subgroup of patients.
00:00

Supplemental Iron
Both oral and IV supplementation are available. As I will show you in the next few slides, oral replacement really does not meet the needs of this subgroup of patients. Iron dextran, iron gluconate and iron sucrose are now available for IV replacement. I will touch on these individually, as the side effect profile, particularly of iron dextran, has more or less been eliminated with the newer IV versions.
00:00
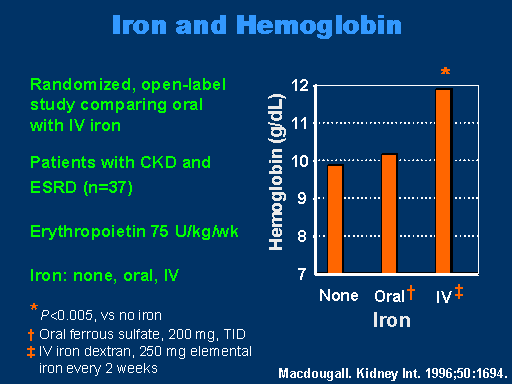
Iron and Hemoglobin
MacDougall showed in this study looking at iron and hemoglobin levels that when you compare no iron, oral iron and IV iron with a fixed dose of epoetin alfa at 75 units per kilo per week, you have a significant increase with IV iron.
00:00
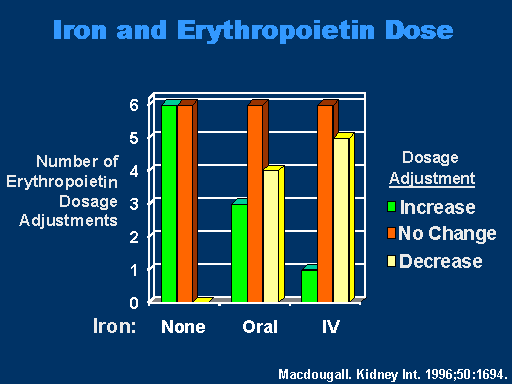
Iron and Erythropoietin Dose
Looking at this another way, if you look at the dose adjustments of erythropoietin with no iron, oral iron and IV iron, what you see here is that as you move to use IV iron, the number of decreasing doses increases. In other words, the requirements of erythropoietin decrease as compared to no iron or oral iron.
00:00

Impact of IV Iron on Erythropoietin
What about the impact of IV iron on erythropoietin? Two studies, both by Park and Senger, showed that dose reductions of 21-77% result in cost savings of $500 to $5,000 in this population. So certainly replacement of iron deficits is important.
00:00
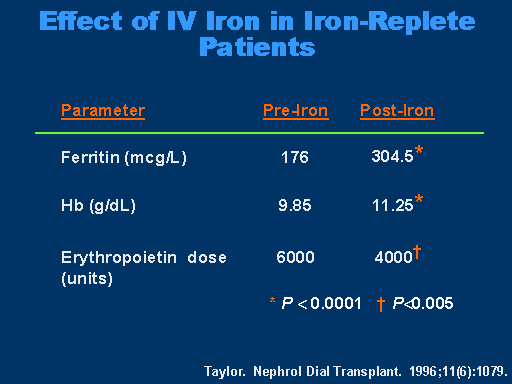
Effects of IV Iron in Iron Replete Patients
Just taking this one step further, the use of iron in iron-replete patients. There are some data out there suggesting that there may be problems associated with this as far as infections. But at least in this study, Taylor showed that in iron replete patients, you can increase the ferritin levels almost double from 176 to 304. You get a nice response into the target range of your hemoglobin, and drop the dose of erythropoietin with the subsequent cost savings.
00:00

Once Weekly Dosing of Epoetin Alfa: Overview
Moving away from iron replacement, let me talk for a moment on dosing. Most of our experience and most of your experience in dosing has been garnered in the dialysis end-stage population. Zappacosta, Graf, and Yagil have presented some data in the last 10 years suggesting that weekly dosing in CKD is effective. Indeed, there is currently a large ongoing prospective study by Bill Owen and myself looking at weekly dosing. I will share some of that information with you. But much of the information that we have in this area has come from our colleagues in oncology and infectious disease in HIV and cancer patients. As I'll show you, the status shows that treatment with weekly dosing is both effective and has a positive impact on quality of life in this population.
00:00
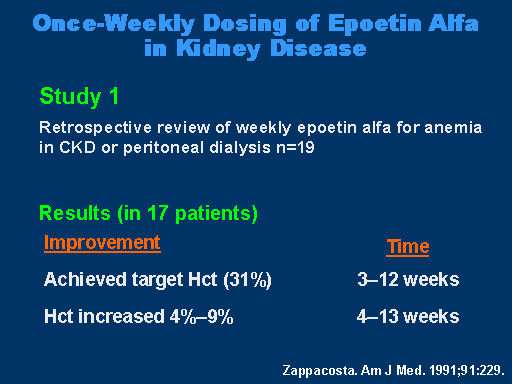
Once Weekly Dosing of Epoetin Alfa in Kidney Disease
As I mentioned, Zappacosta showed in a small retrospective study of using weekly epoetin alfa for anemia and CKD that there was an improvement -- this was in '91, I believe -- in the target hematocrit in 3-12 weeks in 17 out of 19 patients. The hematocrit increased from 4% to 9%.
00:00

Once Weekly Dosing of Epoetin Alfa in Kidney Disease (cont.)
Graf has similar results in an open label study of CKD patients. It was a much larger study, 123 individuals, with a baseline hemoglobin of less than 10, the initial dose of epoetin alfa of 10,000 units subcutaneously weekly. His target hemoglobin was 11-12, and the duration of the study was 3 months.
His results showed that within a month the hemoglobin raised to the target area, that the dose of epoetin alfa dropped to 9,000, and more important -- and this was, I think, in '92 -- there was no deterioration of renal function, which was a concern.
00:00

Once Weekly Dosing of Epoetin Alfa in Kidney Disease (cont.)
Yagil had similar results in an open label study, dosing 4,000 units a week with a target of 10-12, duration of 24 weeks. Indeed, anemia was treated, the maintenance dose dropped, and there was no deterioration in renal function.
00:00

Once Weekly Dosing: POWER Study
Taking this information, Bill Owen initiated a prospective study of patients with CKD and anemia with hemoglobins less than 10. This is currently still enrolling patients. The population target size is 1,500. All patients were started on 10,000 units subcutaneously as initial therapy, and official parameters of quality of life, hemoglobin, hematocrit, transfusion requirements and adverse effects were looked at.
00:00

POWER Study Preliminary Results
Preliminary data that were presented yesterday in a poster session with 615 patients currently being analyzed, showed a 92% response rate over 16 weeks with an increased hemoglobin with a mean of 2.7 grams per deciliter. All quality of life parameters increased significantly from baseline. Although not shown here, transfusion requirements were essentially eliminated.
00:00

Optimal Dosing: Other Disease States
As I mentioned, much of the data on weekly dosing were garnered from the oncology field. I'm just going to touch very briefly on this. The data of once-weekly dosing showed both the treatment of anemia and improved quality of life is possible with weekly dosing.
00:00

Once Weekly Dosing of Epoetin Alfa in Oncology
Four studies looking at weekly dosing, and then three-times-a-week dosing. These two studies use 40,000 a week. These studies used 3,000 units 3 times a week. So essentially, 9,000-10,000 units a week.
The bottom line here is that this was effective, bringing the hemoglobins into target range with a mean change of anywhere from 1.8 to 2.5 grams per deciliter.
00:00
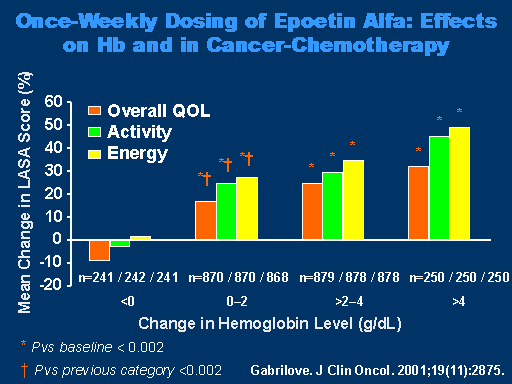
Once Weekly Dosing of Epoetin Alfa: Effects on Hb and in Cancer Chemotherapy
Looking at once-weekly dosing, again, in cancer chemotherapy patients, as you can see here, both the quality of life, the activity of these patients and their energy levels increased as you had an increasing change in the hemoglobin from essentially no change to greater than 4 grams per deciliter.
00:00

Weekly Dosing: Epoetin Alfa in HIV
Some data looking at HIV patients. Again, Saag and Wasserman showed increasing from their baseline anemia values of 9.76 and 11 to 12 and 13, respectively, with a mean change of approximately 2.5 grams per deciliter and a marked improvement of quality of life in Saag's study.
00:00

Optimal Dosing: Epoetin Alfa
But what is optimal dosing? Once-weekly dosing has been shown to be safe and effective in the studies that I had just presented to you. There is a prompt hemoglobin response into the target range at approximately one month, and there is an improvement of quality of life. I'm not going to try to go back on this, but dosing convenience is important, and the logistics of dosing in a busy CKD clinic, in your practice, is also important. Not only is dosing convenience an issue, but there are ongoing studies now looking at dosing every other week, every third week and every fourth week, not only to help improve patient compliance, but to help the logistics in a busy practice. These data would probably be available certainly for the next ASN, but is a direction that is being looked at now.
In summary, anemia is prevalent, as pointed out earlier, in patients with CKD and is currently under-treated. We know that currently anywhere from 30-50% of patients with anemia presenting to nephrologists are not treated. Iron supplementation is extremely important, and there are many forms of IV therapy now that not only makes therapy with epoetin alfa more effective clinically, but more cost effective.
There are adequate data out there now with once-weekly dosing of epoetin alfa, which shows that it is effective and improves quality of life. As I mentioned, additional dosing regimens are out there. They are currently investigational, but there are data suggesting that this is something that will be available soon. Thank you.
References
1. No authors listed, IV. NKF-K/DOQI Clinical Practice Guidelines for Anemia of Chronic Kidney Disease: update 2000. Am J Kidney Dis. 2001 Jan;37(1 Suppl 1):S182-238. No abstract available.
2. Macdougall IC. Is high peritoneal transport rate an independent risk factor for CAPD mortality? Curr Opin Hematol. 1999 May;6(3):121-6. Review.
3. Goodnough LT, Skikne B, Brugnara C. Erythropoietin, iron, and erythropoiesis. Blood. 2000 Aug 1;96(3):823-33. Review.
4. Macdougall IC, Tucker B, Thompson J, Tomson CR, Baker LR, Raine AE. A randomized controlled study of iron supplementation in patients treated with erythropoietin. Kidney Int. 1996 Nov;50(5):1694-9.
5. Park L, Uhthoff T, Tierney M, Nadler S. Effect of an intravenous iron dextran regimen on iron stores, hemoglobin, and erythropoietin requirements in hemodialysis patients. Am J Kidney Dis. 1998 May;31(5):835-40.
6. Zappacosta AR, Perras ST, Bell A. Weekly subcutaneous recombinant human erythropoietin corrects anemia of progressive renal failure. Am J Med. 1991 Sep;91(3):229-32.
7. No authors listed. Effectiveness and safety of recombinant human erythropoietin in predialysis patients. Austrian Multicenter Study Group of r-HuEPO in Predialysis Patients. Nephron. 1992;61(4):399-403.
8. Yagil Y. Proposed therapeutic algorithm for the treatment of anemia of chronic renal failure in pre-dialysis patients with low dose once weekly subcutaneous r-HuEPO. Multicenter Study Group, Israel. Isr J Med Sci. 1997 Jan;33(1):36-44.
9. Gabrilove JL, Cleeland CS, Livingston RB, Sarokhan B, Winer E, Einhorn LH. Clinical evaluation of once-weekly dosing of epoetin alfa in chemotherapy patients: improvements in hemoglobin and quality of life are similar to three-times-weekly dosing. J Clin Oncol. 2001 Jun 1;19(11):2875-82.
10. Demetri GD, Kris M, Wade J, Degos L, Cella D. Quality-of-life benefit in chemotherapy patients treated with epoetin alfa is independent of disease response or tumor type: results from a prospective community oncology study. Procrit Study Group. J Clin Oncol. 1998 Oct;16(10):3412-25.
11. Glaspy J, Bukowski R, Steinberg D, Taylor C, Tchekmedyian S, Vadhan-Raj S. Impact of therapy with epoetin alfa on clinical outcomes in patients with nonmyeloid malignancies during cancer chemotherapy in community oncology practice. Procrit Study Group. J Clin Oncol. 1997 Mar;15(3):1218-34.
12. Gabrilove JL, Cleeland CS, Livingston RB, Sarokhan B, Winer E, Einhorn LH. Clinical evaluation of once-weekly dosing of epoetin alfa in chemotherapy patients: improvements in hemoglobin and quality of life are similar to three-times-weekly dosing. J Clin Oncol. 2001 Jun 1;19(11):2875-82.
| This educational activity is supported by an educational grant from Ortho Biotech Products, L.P. This activity has been planned and produced in accordance with the ACCME Essential Areas and Policies. This educational activity is based on an ASN/ISN evening symposium which was planned by the World Congress of Nephrology program committee. |
| |||||||||||||||