|
|
|
 |
The Advancement of Clinical Practice in Chronic Kidney Disease RPA Satellite Symposium. March 22, 2002 The Interrelationship Between Cardiovascular disease and CKD |
|
00:00
Dr. Patrick Parfrey:
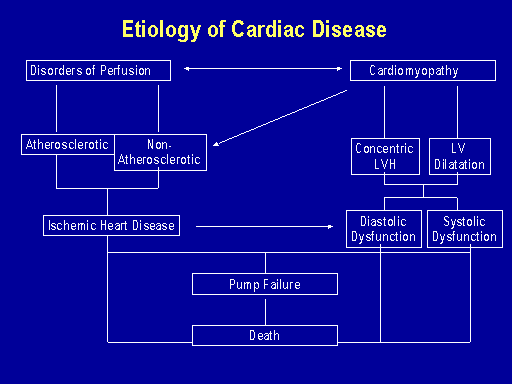
Thank you for the invitation. I'm going to first of all discuss what cardiac conditions occur in chronic kidney disease and what the risk factors are for those conditions and then what can we do about them. I apologize for this first slide because it's not lighting up very well. But you can see simplistically I've divided the causes of cardiac disease into diseases of perfusion which include critical coronary artery disease also known as atherosclerotic disease, non-atherosclerotic disease in which there are non-critical stenoses.
That leads to clinical ischemic heart disease causing angina and myocardial infarction. And then cardiomyopathy which may be caused by left ventricular hypertrophy, LV dilatation, which lead to both diastolic dysfunction and subsequently to this systolic dysfunction. These conditions predispose to pump failure or left ventricular failure which in turn predispose to death. We know from most of the studies that the pump failure is a bigger predictor of mortality than the presence of symptomatic ischemic heart disease in these people.
00:00

Consequences of overload cardiomyopathy
Chronic kidney disease is the quintessential model of left ventricular overload cardiomyopathy. That's because of the high prevalence of things that lead to LV pressure overload including hypertension, arterial sclerosis, occasionally aortic stenosis and also the high prevalence of left ventricular overload. We also usually have hypervolemia and anemia in the predialysis state and then the fistula is added to that, which leads to initially good hypertrophy which subsequently becomes maladaptive and leads to myocyte death.
00:00

Framingham heart study
Now, one of the beliefs of people in treating patients with chronic kidney disease is that this is an atherogenic state. I'm going to discuss whether in fact it is an atherogenic state per se or whether it's just a marker for increased risk because of underlying cardiac diseases. The Framingham study investigated 6,233 people enrolled in a community study with 15 years follow-up. Of those people in the study about eight percent had a raised serum creatinine at these levels for men and for women, and of those people with renal insufficiency 19 percent had cardiovascular disease.
00:00
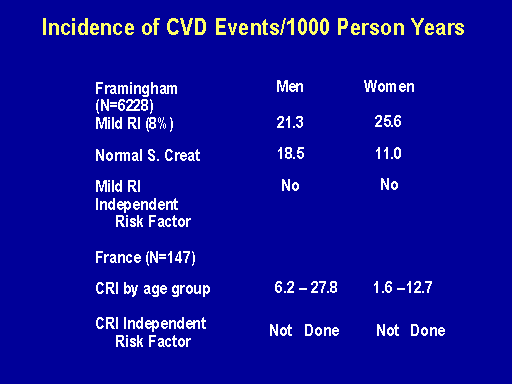
Incidence of CVD events/1000 person years
And what's of interest here is this is the incidence rate of cardiac events per 1,000 patient years for men, which is 21.3, and for women, 25.6 in those with mild renal insufficiency. Whereas those with a normal serum creatinine had quite higher event rates for men and lower event rates for women. But when the increased prevalence of diabetes and hypertension, et cetera, was factored in to these event rates in those with mild renal insufficiency, it was determined that renal insufficiency was not an independent cardiac risk factor, coronary risk factor.
The data that's presented frequently about renal insufficiency being an atherogenic state was this data from France in which chronic renal insufficiency patients were presented by age group with event rates going from 6.2 to 27.8, which is very similar to the event rate up here and up here. And there was no multivariate modeling done to demonstrate that renal insufficiency was actually an independent risk factor.
00:00

Hypertension detection and follow up program
Now, the Framingham data has been confirmed by the NHANES data and by the MRFIT data that renal insufficiency in community studies is not a risk factor for subsequent primary events. However, in cohort studies where there's a higher degree of comorbidity such as this hypertension detection follow-up program in which 11,000 people were enrolled, there was clear evidence for increased cardiovascular mortality, three fold higher in those with a raised serum creatinine than those without. Similarly this evidence is being confirmed in studies of the elderly and in studies of coronary patients, cardiac patients.
00:00

Interpretation
The HOPE study as well was an important study that demonstrated that renal insufficiency was an independent risk factor for subsequent cardiac events. However, the interpretation may well be that in cohorts of hypertensives, older people and primary patients where the baseline comorbidity is quite high, that renal insufficiency may be a marker for already established, more severe generalized vascular disease and not a risk factor for accelerated atherosclerosis causing cardiac death.
From the perspective of physicians knowing that renal insufficiency marks for subsequent adverse events in cohorts with high comorbidity that's important because we can target them for intervention. But for people who are examining the mechanisms of atherogenesis and chronic renal insufficiency, if it's not a risk factor then maybe they're barking up the wrong tree. Now, if we look at the risk factors that we know about in chronic kidney disease for cardiac events let's first of all look at left ventricular hypertrophy then I'll look at cardiac failure and then I'll look at ischemic heart disease.
00:00

Baseline characteristics
So this is a Canadian study published in '99 of between 200-300 patients whose mean creatinine clearance was 37 ml per minute, whose baseline hemoglobins were 12.8 and blood pressure 144 over 85. So they didn't have severe risk factors.
00:00
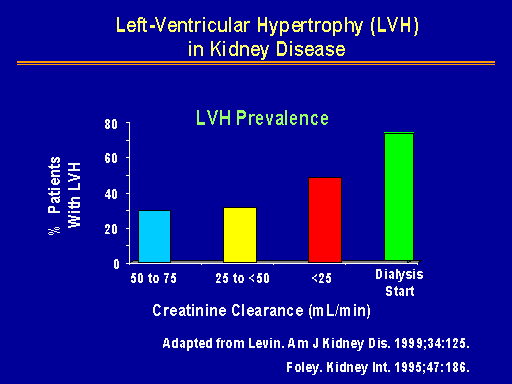
Left-ventricular hypertrophy in kidney disease
The Canadian investigators demonstrated that the prevalence of left ventricular hypertrophy varied by the degree of renal impairment, rising as the GFR fell as you can see here.
00:00
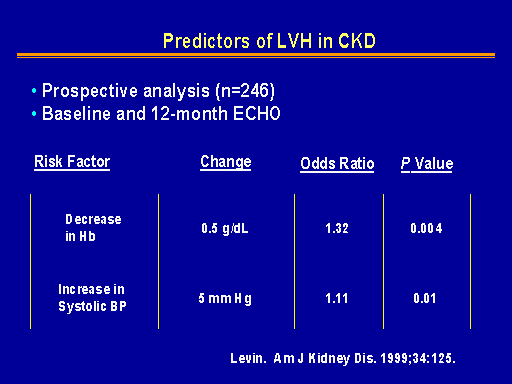
Predictors of LVH in CKD
And the risk factors that were identified for increased left ventricular growth defined as a 20 percent increase or 20 grams per square meter increase were anemia, in which there was a 30 percent increased risk for LV growth per 0.5 grams per deciliter decrease in hemoglobin from normal. Blood pressure was also seen to be a risk factor. There was a 10 percent increase in LV growth for each 5 millimeter of mercury rise above the normal level and that was independent of the underlying left ventricular mass index.
00:00

Baseline characteristics of 638 renal transplants
Now, this data that I'm going to show you is in press in JASN and what it examines is 638 renal transplant recipients who have been transplanted for one year and they're in fact every patient transplanted in two Canadian provinces, Manitoba and Newfoundland. These renal transplants had no cardiac disease at the start of the study that we did. Here are the baseline characteristics at one year: the majority were on cyclosporin, blood pressure as 138 over 85, creatinine was 160 micromoles per liter which is about 1.8 milligrams per deciliter according to what you have in the United States. The estimate creatinine clearance was 55 ml per minute, the hemoglobin was 126 grams per liter and the albumin was 39 grams per liter.
00:00

Incidence of de novo heart failure
And the mean follow-up was 7.2 years. During that period there was an incidence of 1.26 events per 100 patient years of de novo heart failure. That's about five times greater than occurs in the general population.
00:00

Risk factors for de novo heart failure
And the risk factors identified for de novo heart failure included, independent of age and diabetes, these potentially reversible risk factors: anemia, 24 percent increased risk per 10 grams per liter decrease; systolic blood pressure, 29 percent increase per ten millimeters of mercury increase; and hypoalbuminemia, a 110 percent increase per gram per liter decrease in the serum albumin.
00:00
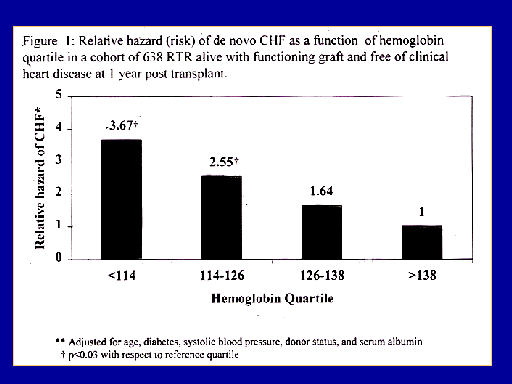 |
Hemoglobin vs. relative hazard of CHF
Now, that might be familiar to you from other papers and I'll briefly discuss that later on. This shows you the dose response curve that was resolved for the fall in hemoglobin from a normal value of 138 g/l and the hazard ratio for the subsequent development of de novo heart failure. You can see that even a fall down to 126 led to a 64 g/l percent increased risk of de novo heart failure. And that risk decreased, going down to 114 to 126 and the risk was as high as 3.7 times higher if the hemoglobin was below 114 grams per liter. Striking evidence for the impact of anemia on the development of de novo heart failure. These slides have only recently been put together so that's why they're a poor quality here.
00:00
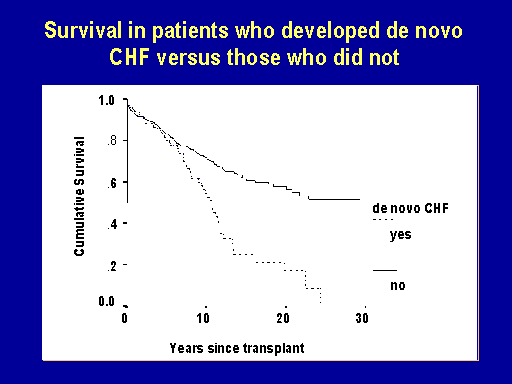
Survival in patients who developed de novo CHF versus those who did not
This demonstrates that those people who developed de novo heart failure had a worse outcome than those people who never developed de novo heart failure.
00:00

Incidence of de novo ischemic heart disease
What about the evidence for ischemic heart disease? The event rate was identical for those people who developed angina or myocardial infarction to that of the development of de novo heart failure, identical. And that's not what happens in the general population. You can see that that event rate is identical to the Framingham data, both for the general population and for the people with renal insufficiency. I should say similar, not identical.
00:00

Risk factors for de novo ischemic heart failure
The risk factors for de novo heart failure independent of age, diabetes and gender, included high blood pressure and allograft rejection but did not include anemia.
00:00
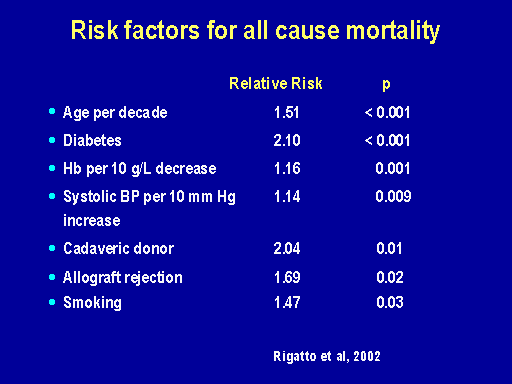
Risk factors for all cause mortality
The risk factors for all cause mortality that were reversible included anemia, high blood pressure again and smoking.
Now, it's been clearly said by everybody and will continue to be that the opportunity for risk factor intervention occurs in the chronic kidney disease stage before dialysis. Because when starting dialysis in Canada here's the data which was collected in a prospective study where the doctors examined the patients the day they started dialysis. Thirty-five percent of people had heart failure, a further 80 percent had myocardial infarction and 21 percent had angina. Strikingly similar, that prevalence is, to the data that Bill McClellan just presented us.
00:00
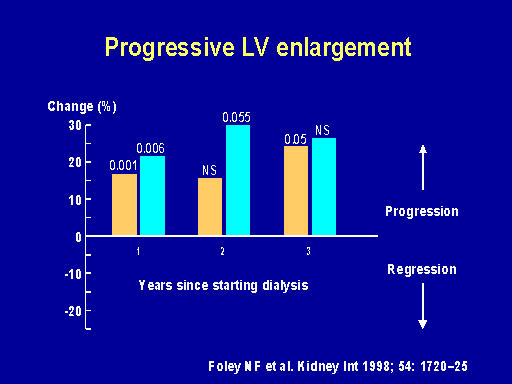
Progressive LV enlargement
And just a quick review of what happens when people start dialysis. There's a deterioration in the left ventricular volume and left ventricular mass index that occurs over the following few years that's associated with being on hemo rather than PD and is associated with anemia.
00:00

Independent risk factors for CHF
And then when you compare the development of de novo heart failure in people who develop it after starting dialysis compared to those people who never develop heart failure while on dialysis, the risk factors are very similar to those observed in that transplant group, independent of age, diabetes and the underlying echo: anemia, hypoalbuminemia and high blood pressure. And for ischemic disease the risk factors again independent of age, diabetes and the smoking were high blood pressure and hypoalbuminemia.
So what's interesting is the reversible risk factors here in the various chronic renal disease populations. We've got those ones that have chronic renal insufficiency I suppose you might say, hypertension and anemia associated with left ventricular hypertrophy. In dialysis patients it's the hypertrophy associated with being on hemo rather than PD, high blood pressure and anemia.
Heart failure was associated with hypertrophy, hypertension, anemia and low albumin. Ischemic heart disease is associated with hypertrophy, hypertension, low albumin, lipids, but not anemia. In the transplant group high blood pressure associated with hypertrophy, anemia, hypertension, low albumin, delayed graft function for heart failure. Hypertension, lipids, acute rejection for ischemic heart disease. Now, what's interesting here is that commonality across every population for two major hemodynamic risk factors: high blood pressure and anemia. Very, very consistent.
00:00
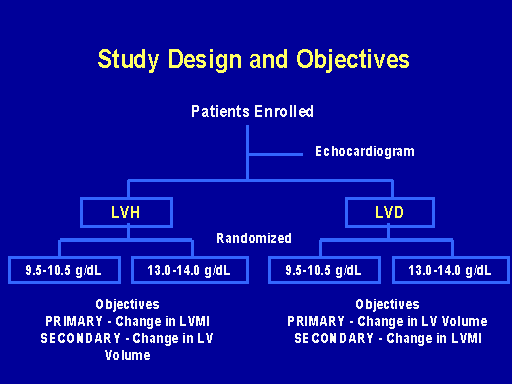
Study design and objectives
I'm going to briefly now just discuss the issues of intervention. Clearly we have reversible risk factors demonstrated in properly designed longitudinal studies, but we do not have that many studies investigating chronic kidney disease patients for some of these risk factors, particularly anemia. As Bill said there are multiple interventions that are efficacious in preventing progression and it looks like as if losartan and ACE inhibitors can prevent cardiac events so therefore they should be used properly.
The anemia story is difficult because we have no clinical trials. This is one of dialysis patients in which we looked at the outcome for people who had left ventricular hypertrophy or LV dilatation who were normalized to hemoglobin normal or partially corrected.
00:00
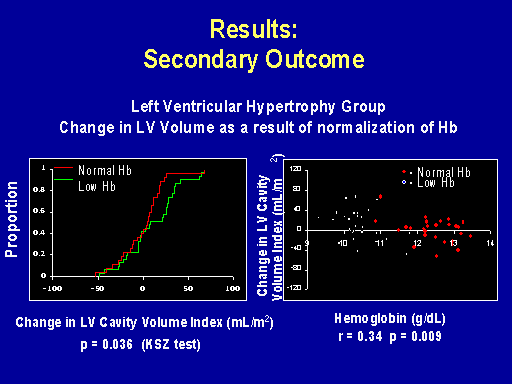
Results: Secondary outcome
And I'll just quickly show the data in that there appeared to be prevention of left ventricular dilatation. You can see here there's no dilatation having occurred in the intervention group compared to the control group.
Here's the relationship between the change in left ventricular volume over one year compared to the achieved hemoglobin in that trial. And you can see there's a significant relationship between the two outcomes. However, that wasn't associated with a significant regression of left ventricular mass index. I suspect that's because there was inadequate statistical power.
00:00
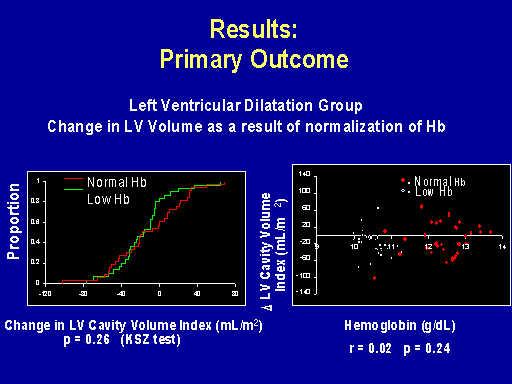
Results: Primary outcome
In patients who had severe LV dilatation, you can see here that in the control group there was some degree of regression of left ventricular volume but there was no difference to the intervention group.
00:00

Normalization of hemoglobin
So we concluded from that trial that normalization of hemoglobin does not induce regression of concentric hypertrophy or LV dilatation. It prevents progressive LV dilatation in patients with normal left ventricular volumes and it may improve some dimensions of quality of life. So taken together with the Amgen study we felt that the utilization of erythropoietin to normalize the hemoglobin level should be tested in patients at the earlier phases of their cardiac disease. And such studies are in progress including one in patients starting dialysis, a large study which will be reported next year which has got echo outcomes and quality of life outcomes. Donal Reddan is going to discuss the American study that's planned and there are some studies in Europe that are investigating this.
In Canada, we're planning to do a multiple risk factor intervention study of patients who have got moderately severe renal insufficiency in which those interventions will be applied by a nurse and a clinic, supervised by a nephrologist according to preplanned algorithms, to determine whether that intervention is more efficacious in preventing renal events and in preventing cardiac events than conventional therapy that's directed by the family doctor who uses his consultants as he sees appropriate in which there will be about two and a half thousand patients enrolled. So that's it. Thank you.
Discussion:
Audience member: It seems that there is a contradiction in the way the serum albumin being low is a risk factor. So it means basically we have at least to normalize it. On the other hand, we have to restrict protein in these patients who have renal failure to preserve their kidney function.
Dr. Patrick Parfrey: I think the current recommendations would be that it would be moderate protein restriction which is unlikely to lead to low albumin levels. And I think the question for us would be what is it that low albumins are marking for that is causing heart failure and causing ischemic heart disease, because it could be different things. It could be a reflection of malnutrition, or it could be a reflection of inflammation, or it could be a reflection of other things that we're not sure about. Maybe it's linked to higher homocysteine levels or other atherogenic risk factors. I think that part of the equation needs to be worked out mechanistically.
Audience member: In the transplant study, cadaveric transplants seemed to be a risk factor. Was that related to the renal function of the cadaver versus living related transplants? Or do you have any other explanation?
Dr. Patrick Parfrey: We looked at the transplants in particular because we thought that it was a model of renal insufficiency which was associated with immunosuppression use, that's why we were interested in it. We didn't find an independent effect of the glomerular filtration rate over and above the risk factors that we already had information on for either of the cardiac events, heart failure or ischemic heart disease. What it is the cadaveric donor is marking for besides those risk factors, I don't know. But it doesn't seem to be something to do with the GFR and it doesn't seem to be something to do with the immunosuppression as far as we can judge in a study that's got 638 patients. Because as you can imagine that's not really that big a study when it comes down to working out the role of smaller risk factors.
Dr. Anton Schoolwerth: Thank you, Pat. We probably should go ahead.
References
1. Culleton BF, Larson MG, Wilson PW, Evans JC, Parfrey PS, Levy D. Cardiovascular disease and mortality in a community-based cohort with mild renal insufficiency. Kidney Int. 1999 Dec;56(6):2214-9.
2. Shulman NB, Ford CE, Hall WD, Blaufox MD, Simon D, Langford HG, Schneider KA. Prognostic value of serum creatinine and effect of treatment of hypertension on renal function. Results from the hypertension detection and follow-up program. The Hypertension Detection and Follow-up Program Cooperative Group. Hypertension. 1989 May;13(5 Suppl):I80-93.
3. Levin A, Thompson CR, Ethier J, Carlisle EJ, Tobe S, Mendelssohn D, Burgess E, Jindal K, Barrett B, Singer J, Djurdjev O. Left ventricular mass index increase in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis. 1999 Jul;34(1):125-34.
4. Foley RN, Parfrey PS, Harnett JD, Kent GM, Martin CJ, Murray DC, Barre PE. Clinical and echocardiographic disease in patients starting end-stage renal disease therapy. Kidney Int. 1995 Jan;47(1):186-92.
5. Rigatto C, Parfrey P, Foley R, Negrijn C, Tribula C, Jeffery J. Correction of acidosis in hemodialysis patients increases the sensitivity of the parathyroid glands to calcium. J Am Soc Nephrol. 2002 Apr;13(4):1084-90.
6. Foley RN, Parfrey PS, Kent GM, Harnett JD, Murray DC, Barre PE. Long-term evolution of cardiomyopathy in dialysis patients. Kidney Int. 1998 Nov;54(5):1720-5.
| This educational activity is supported by an educational grant from Ortho Biotech Products, L.P. This activity has been planned and produced in accordance with the ACCME Essential Areas and Policies and with CE policies of the State of California Board of Registered Nursing. From a CME Symposium held on March 22, 2002 at the Renal Physicians Association (RPA) Annual Meeting in Washington, DC. This symposium was approved by the RPA. It was not part of the official RPA Annual Meeting as planned by the RPA Program Committee. |
| |||||||||||||||