|
|
|
 |
Selected Talks
National Kidney Foundation Spring Clinical Meetings, Dallas, TX, April, 2003 System Barriers to Creating the Best Angioaccess for Our Patients |
|
| ||||||||||||||||||||||||||||||
Moderator: : Jeff is going to talk about system barriers to creating the best angioaccess. Dr. Sands has a unique background that I suspect that most of the audience is aware of. He was a nephrologist in the trenches like most of us for many years in Scranton, Pennsylvania. I think he wrote some of the definitive articles in terms of access monitoring using ultrasound. For the last three years he has been with Fresenius Medical Care as Vice President of Medical Technologies. His topic, I think, is very pertinent to this particular area. I think also something that we should know about Jeff; he actually lives in Celebration, Florida right outside of Walt Disney World and has a year-long pass to Disney World, which I think is unique in and of itself. Jeff, we look forward to your talk about the system problems that we encounter in increasing fistulas in the U.S.
00:00

Dr. Jeff Sands:
Thanks very much. What Jack has asked me to do is set the stage for the later talks and talk to you about the system of vascular access care, what works, what doesn't work and really not go through a lot of data. Normally I would go through a lot of data. And the purpose of this is really to move on and let Allan and Jack talk about some of the specifics.
00:00

System of Vascular Access Care
So what I would like to talk about is the system of vascular access care, where we are, what is the morbidity, the mortality, the cost; talk about the cycle of access placement with repair and loss and then move on to talk about some of the opportunities and barriers. I am not going to tell you anything you do not know; what I want to do is put it all in a framework so that we have all the things on the table at the same time.
00:00

Why focus on vascular access?
Why are we talking about vascular access? Why do we care? The reason is because, as everybody in the room is aware, access problems in procedures are at epidemic proportions. We are doing over one procedure per patient here, numbers of declots revisions, and we have an epidemic of catheters, sepsis, hospitalization and death. It is what we all know; it is a disaster out there.
00:00

US & European vascular access
Now, if we look at the background data - and this is a summary slide from some of the DOPPS work - what we can do is compare where we are and where Europe is and talk about it a little bit. Looking at the access used for the very first dialysis ever, 67% of European DOPPS centers start with a fistula. Fifty-nine percent of the US centers start with a catheter. If you look at prevalent patients, 81% of the Europeans had fistulas; 24% of the Americans. Look at our graft rate and look at our catheter rate. If you just look at that, that sets up the problem; you do not have to go any further.If you look at the life tables, fistulas in Europe do better than fistulas in the US, and both of them do better than grafts. And if you look at the relative risk of needing a declot or a revision it is seven-fold higher if you have a graft and a fistula, and a procedure is eight-fold higher. So inherently the problem that we are faced with is a problem of grafts and catheters. Starting up front, no matter how good you are, no matter how wonderful your care is and no matter how wonderful your team is, if you have a lot of grafts and you have a lot of catheters you are doomed to failure.
00:00

Access type and mortality risk
Now, how does that affect mortality risk? This is Dhingra's work and what you can look at is prevalent and incident patients, non-diabetics. Look at the catheter patients; look at the relative risk of death, 70% higher. Incident patients a little bit. If you look at the diabetics, again, catheter patients, graft patients have a much higher risk of death. So not only are we causing procedures and multiple interventions, but the people are dropping dead.
00:00

This is some of Dr. Lowrie's data from the Fresenius Medical Care (FMC) system. What this does is the same kind of analysis only the other one was only adjusted for case mix. This is adjusted, if you look in the red panel, for case mix and every bit of lab data, demographic data that we keep: Kt/V, fatty mass index, whether or not you are a diabetic, what your albumin is, and all those things. The relative risk if you have death, if you have a catheter, controlled for everything is 1.95. And if you have a graft it is 1.32. So inherently we are going to have bad outcomes.
Now if I move along, there are a lot of ways to deal with the catheter epidemic because we are now in a world where, as I go around the country, it is very common to see 30% catheters; I have been in units with 60% catheters. Some of them do okay for a while, some of them have horrible outcomes. There are a lot of ways to deal with catheters. One way is to get rid of them. The second way is to see if you can do something about the morbidity and mortality associated with them.
00:00

Polysporin & catheter morbidity
This is from Charlene Lok; a randomized trial from Toronto looking at what happened when they used polysporin in a randomized, blinded controlled trial, on the exit site. Look what happened to bacteremia rates, look what happened to survival rates. Infections dropped from 4.1 to 1.02 per thousand catheter days. Bacteremia dropped, catheter removal dropped and the whole gamut. So there may be a bunch of approaches, but clearly what is going on in the catheter system is very unforgiving. And so, very small breaks in the process that you might not be able to figure out cause very bad outcomes.
00:00

Practice Pattern Implications
What does that mean to people? What it means in your unit is you have got an epidemic of disasters. If you take that and put it in a spreadsheet you assume 2.3 bacteremias per thousand catheter days and that is about what you see in the literature. About half of it is Staph. aureus; major complication about a quarter. Fourteen to forty-four percent have serious metastatic complications. There is about a 16.5% death rate. What that means is you have somewhere between 3000 and 8000 preventable deaths or major complications in a population of about 80,000 patients.In any other field but nephrology that would be intolerable. To what patient are you going to say, that you are going to use a system where 10% of you are going to be gone next year, because we are not going to be bothered with what is going on; and for a bunch of the rest of you, we will need to give you (heart) valves, or we may need to deal with an epidural abscess? This is not effective and it is horrible for patients and everybody in the room knows it and deals with it.
00:00

K/DOQI clinical practice guidelines for vascular access
The real interesting question now when I look at this is: Here we are eight years after DOQI and a wonderful document that talked about fistula placement, catheter reduction, location, surveillance, intervention. And we can argue about some of the details, but the DOQI team did a great job. The real question is, why hasn't the care gotten better? You have the guideline; everybody pretty much agrees, and if you look on the ground it just flat-out isn't happening.
00:00

Why hasn't care improved?
So why has the care not improved? Well, the first issue is that the focus has been only on dialysis facilities. And the greatest improvements are from decreasing catheters and increasing fistulas. That involves vascular surgeons, hospitals and nephrologists. There is not one nurse in one dialysis unit who can write the orders to get this stuff done. And again, when you try to change things it is a lot easier to change things from an upstream problem if you deal with it upstream, not downstream.Lack of accountability: If you go into hospitals, to many surgeons, interventional radiologists, a lot of them come in and do not even know what their outcome data is. How do you deal with outcomes if nobody knows what you are looking at? Everybody thinks they do a great job; maybe they do, maybe they don't.
Financial disincentives: Physicians, hospitals and device manufacturers have incentives built in the system not to do the right thing. Now, dialysis facilities on the other hand are much more aligned with the patient. If you send a catheter patient to a dialysis facility we lose money. We are much better off without a catheter. So, the system creates or rewards behavior that may not be what we choose or want.
Educational deficit: Some people just flat-out do not know.
00:00

Financial disincentives
Now just to illustrate financial disincentives, this slide is a little old so it is not exactly accurate now but it is close. If you look at what Medicare pays the physicians for fistula placement it is $573 in Northeast Pennsylvania. Transposed fistula $816 - now we are talking real surgery; a couple of hours in the OR, you could really hurt somebody, right? That is not a whole lot of money for that. Look at the graft: $759. And look at the revisions. So, grafts become the gifts that keep giving. They are an annuity: If you put some in you get to do a whole lot of work. Look at catheter placement; what is that really? That makes the patient go away. That is a shift the burden strategy. You have a problem and you just make it go away. Now, that does not really do it with everybody, but people make decisions because of real reasons.
00:00
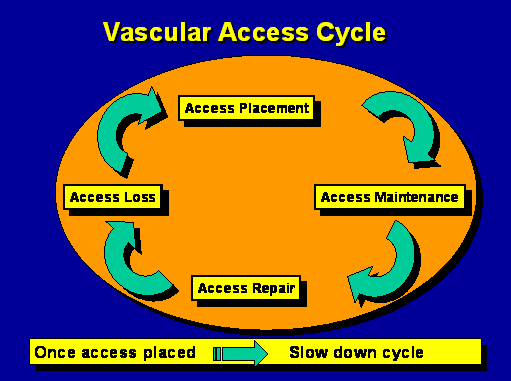
Vascular access cycle
When I was making rounds and I was in practice I knew that at 1:00 in the afternoon, if I started at 7:00 I better have the same amount of work at 1:00 in the afternoon that I had at 7:00 in the morning or I was in trouble because my goal in life was to get the right data on the chart. I had to get through everybody before midnight. Well, there is so much time you can spend with every patient. If you have to see ICU patients they take a half hour. There are a certain amount of patients that you can see in a certain amount of time. You start changing your decisions as the day goes on and how you allocate your time because you have got to get the work done. But every time you change your decision and it results in shifting it somewhere else and you wind up with a catheter, you just made a ten or twenty or thirty thousand dollar decision for reasons that may or may not have been appropriate outside of your own context.So when I look at vascular access what I do is I look at it as a cycle where we go from placement to maintenance to repair to loss. And once we place an access, what our goal is is to slow down the cycle. You want to place it and use it forever and never have a problem. So let us talk about that and kind of dissect that cycle and what is involved.
When you talk about placement, first you have got to identify the patient. Then they hopefully go for nephrology in CKD care because if you get them early enough it gives you a lot more room. Then you refer them for an access placement and then they go to another level. The surgeon evaluates them, hopefully they place a fistula, and then there is some kind of follow up.
00:00

Emergency access placement
What happens if you throw a barrier in anywhere here? It does not have to be all of them anywhere. What does that do? It inevitably increases emergency access placement, right? We see it everyday. We see patients who wind up in the hospital whom we saw in our office but never got the access in and for whatever reason: the patient did not go, we did not refer them, the surgeon could not get it together; they get an access that does not work or they just show up in the ER. But either way, you wind up with an emergency. And what does that do? The more emergencies you have, the more catheters you have, the more grafts you have, the fewer fistulas you have. It is in the structure of the delivery system.
00:00

Access placement
Now, if you look at that, where do you change that? Where do you have the most opportunity to improve? Your sweet spots, your leverage point is in CKD care at preoperative imaging. And if you do those you get more fistulas and fewer catheters automatically. Who has the impact here? Who are the decision-makers? A little bit is in primary care, but it is nephrologists, surgeons; it is doctors. It is doctors and hospitals. It is not the dialysis facility.There is a caution: One of the things that is going on is, that the Network is talking about having a fistula creation initiative. Well, if you just go putting in fistulas in everybody and you do not image and you do not use your head, what can happen is you can wind up with a catheter epidemic and you may do more harm than good by having more catheters because having a fistula not work may not kill you, but having a catheter may. And again, this is especially true if you attempt fistula placement without imaging.
00:00

CKD care and AVF placement
Now, what is the data for this? This is some of the data from Network 1, looking at first access used ever, factored by when a patient saw a nephrologist. And you can see if you were followed by a nephrologist and had gradual, predictable progression to ESRD, 46% of those folks in New England started with a fistula. If your progression was more rapid and less predictable, 21% had a fistula. And if you showed up acutely it was 2% and look at the catheter rates. All the studies - and there are a number of them - all show the same results.
00:00

Preoperative imaging for AVF placement
Preoperative imaging: This is Silva's paper and there are seven or eight other papers that show the same thing. If you do preoperative mapping, you can go from a situation where the fistula rate in his hands in Newark went from 14% to 63% by moving to mapping; looking for the artery, looking for the vein, having size and Doppler criteria. The same thing you do with carotids, the same thing you do with legs, the same thing you do with any kind of vascular surgery. Do you need to do it every time? Maybe not. If you have big pipes going up their arm and great arteries you probably do not have to. I will let Allen comment on it, but I doubt he does very much surgery without looking. Look what happens to early failure when you do that: it goes way down.What is the secret issue with fistulas? It is what we all know, which is a third of them never work. A third of the fistulas do not work. A third of the fistulas do not work here, a third of the fistulas never work in Europe. If you map and you do some preoperative evaluation, you can get that number way down.
00:00

Successful AVF creation
This is from a paper by Gibson et al. Look what happens when you can do the whole thing. Now granted, this was done in a VA and everybody was male which makes a difference. But it is a nice slide and it really shows what you can do and I think Tony Besarab has shown similar results from his experience in West Virginia where he was able to get the fistula rate up in the 90% range. Getting to 50% is trivial. If you try you can get to 50%. To get to 90% you have to do some work. And there are places where your population will not allow it, but on the aggregate you can get much higher than you think if you try.
00:00

Cost of access insertion
What does that mean for cost if you follow those leverage points, predictably? This is from Allan Collins' data. Look at the difference in the cost of placing an access as an outpatient and as an inpatient. There is only a $9,000 difference. Why waste $9,000 because you did not plan? That is crazy. If there was a finder's fee that paid a nephrologist $4,000 cash for every patient who started with a fistula, do you think we would have more fistulas?
00:00

Access maintenance procedures
All right, let us move along and talk about maintenance. Again, the same kind of thing: You do access monitoring, that leads to imaging in a select population and elective interventions. And on a second level some of these folks need surgical evaluation, elective repair and follow-up. Again, you flip the barriers in it and numerous barriers for this. And what do you wind up with? You wind up with more emergencies. Again, predictable interventions as an outpatient are a whole lot better for the patients and a whole lot better in terms of cost. Not to mention the problem in a dialysis facility where you pin down your best people trying to make arrangements to deal with stupidity that could have been prevented. What happens when you have those? People wind up in jail, they have procedures, and they have catheters. It is inevitable; it is in the structure of the system.
00:00

Access maintenance
Now, where are the leverage points? Well, the ones I chose were monitoring and elective revision because if you do that you get fewer emergency procedures. You get fewer catheters and fewer total procedures. There are some caveats with that. Who impacts it? Well, usually there are a lot of people who can impact it. But again, reimbursement is key here because none of this activity is paid for. Again, who has the impact? Nephrologists, vascular surgeons, interventionalists, the hospitals. Dialysis facilities are involved, but clearly are not the only focus by any means and not the greatest focus. Now, what is the caution? Technique, population and speed are crucial. If you do not do a monitoring program properly, targeted properly and use your head in this area, all you do is you make work. You do procedure after procedure after procedure. It is not like it was when I started and Tony started doing this 15 years ago. It is very different. Fifteen years ago the incidence of access failure was way higher in grafts and the type of revisions and surgery we did were different. We will talk about that.
00:00

Monitoring & intervention
Where are the data to support this? This is McCarley's study and again, the reason I show this is it is easy to show. This is a sequential study; in this section they did nothing. Here they did dynamic venous pressure, which every paper except one has shown is worthless. And in phase III they did access flow monthly, which you see has a marked decrease in thrombosis in grafts, decrease in fistulas, and an increase in angioplasty work. This is not a perfectly clean study because in a sequential study they did not take into account the lag. Once you start doing angioplasty there is a three to four month lag before you see the thrombosis rate go down.
00:00

Prediction of access failure
Now, I should really show Bill Paulson's work because Bill really is the person with Jack who did some nice work on looking at how well you can predict thrombosis. But I had a better slide on this paper, so I chose to show it. And what you can see is this is looking at the receiver-operator curve access flow with the best predictor of 30-day failure. Dynamic and static venous pressures were not very predictive, but the area under the curve even for flow measurement with 0.73. And when you combine them it did not work.So there has been a whole series of people, some of whom are my friends to the right, who said, "Hey, wait a minute. Is there a reason to do this? Is this an appropriate therapy or are we just making a lot of work and not really helping patients?" And I think you have to start to talk about when it is appropriate. And it has to do with prevalence and incidence and again, I am mathematically impaired so I am not going to show you the way Nikolai Krivitski would show you. I am just going to show you what happens.
00:00

Prevalence implications
What happens if you have a test that is 80% sensitive and 80% specific (like our static venous pressure)? And what if your prevalence of disease is 800 cases per 1000? That is the clotting rate now in grafts. What do you get? Well, you are going to get out of 1000 people, 640 positive tests. And of those people, 640 positives, who have the disease and 40 who test positive who do not have the disease, which is a positive predictive value of 94%. That is pretty good. If 94% of the people I send over to Jack for intervention actually have something, I'm pretty happy with that. And I miss a bunch of people, but that is okay; I don't really care so much about my negative predictive value.What happens if I move into a different world? What if my prevalence is 200 cases per 1000? Now, that is the fistula clotting rate. Now it is a little different. Now, 160 out of 1000 people test positive and have disease and 160 test positive and do not have the disease. So, my positive predictive value is 50% and half the folks I am sending over to Jack do not need to be there. That is your real problem. So what your target population is is very important and that has changed, because when I started doing this, when Tony started doing this the clotting rate in grafts was 1.5 to 2.5 per patient year. In that era, if you just tested the people who had (shoes) you found a lot of disease. And if the therapy worked you had a big improvement in outcome.
00:00

Access monitoring goals
In this era you have a target. If you target patients as Jeff did in his patients who are repeated clotters, you are not going to have a big improvement. If your thrombosis rate is very low as in the Canadian paper where they only looked at prevalent patients and thrombosis happens more early than late and their clotting rate in the control group is 0.4, you are not going to have a big outcome improvement. If you are in the rest of the world where the clotting rate is one per patient year, you can help it a whole lot. But again, you have to know what you are doing and if you wait four weeks to do the intervention, half the things that you were going to help are gone. So, there is a whole bunch of stuff going on.What can you achieve? This is what the data says: If you do access flow you get clotting rates between 0.1 and 0.3 in fistulas and 0.2 and 0.5 in grafts. Now, I came from an ultrasound era. That was a whole lot better. When I was able to do ultrasounds, which you cannot do anymore, I always knew flow and I always knew if they had stenosis. And if I wanted to look up the artery I could, but I was not smart enough to. And in that world you never sent anybody over who did not have anything because you saw both. That is not available right now, so now we have to guess flow is pretty good but nothing is perfect and the target population matters.
Importantly, you can reverse the thrombectomy angioplasty ratio. What that means is you substitute elective procedures for emergent procedures. That is very important in a dialysis unit. Like the Monday morning when you come in and you have three call-ins; well, there is nobody to put the patients on and two people are clotted and they are tying up the chairs and now the clock starts. You have three and a half hours to clear that chair. You have got to get those folks out of there.
Now, when I was making rounds, I never wanted to hear about things on Monday morning, right? I only wanted to see people who could not breathe before noon on Monday morning. So if they called me, what did I do? My behavior was "make it go away". That is exactly the wrong decision. You get a call 6:00 on Monday morning when you are in the shower, you make a $10,000 decision that is based on how quickly I get off the phone so I can finish taking a shower. Now, that is what is going on and it does not have to happen.
00:00

Emergency access procedures
How about repair? Identify the pathology, repair the lesions and confirm improvement. Evaluate for fistula, have a plan. If you have a plan on the chart and they call you at 6:00 in the morning, say "Oh, Charlie is clotted. What does it say on the plan?" "Send to surgery" means left brachial fistula next time. That is a whole lot easier. Now you have made a decision that makes sense and you are still in the shower. Follow up monitoring: if you put the barriers in, again same place with emergency procedures that leads back to hospitalization, more procedures, more catheters and fewer fistulas. It is in the structure.
00:00

Access repair
Where are the leverage points? Well, I say it is in confirming the repair because a whole lot of repairs do not solve the problem. Either you repaired something that was not the cause of the problem or you did an angioplasty and it bounced open and then it bounced back; what is called (infensin) clotted back up. And Tom Vesely in some of his work is finding low flow in grafts now for the people who have inflow disease. And most people do not even look for the inflow disease. I did not used to look for it; I had this psychosis. I thought that the fact that none of my patients had legs had nothing to do with the circulation in their arms. It just never occurred to me that atherosclerosis would happen above the waist.Who has the impact? Just doctors; it is just doctors who can have the impact. But who do we beat up on? We beat up on the dialysis facilities and nurses. That is stupid. Again, for impact you have to go to the people who actually do the work.
What is the caution? One of the problems when you make intervention really easy - you know if you just call up down the street and Jack gets the thing fixed in two hours, and I was blessed with that kind of situation - it gets really hard not to use it. And so there is a tendency in the behavior to not think and just send it down the street and what you get is a procedure epidemic if you do not use your head and you do not monitor the appropriateness of what you are doing. The sixth time you do the same thing it probably is not going to work out better than the fifth.
00:00

Patients with sites for AVF placement
Patients with sites for fistulas: This is a study I did with Mike Lazarus. And what we did was arterial venous mapping by Doppler on all comers in a bunch of dialysis facilities in Philadelphia, Atlanta and a couple of other places. We looked at 444 patients and if you look at the catheter patients, 65% of them had ultrasound criteria that suggested that they had suitable vessels for fistulas and at least 80% of those should work by the criteria. And if you looked at grafts it was 73%. And when you looked at flow on loop graphs, 80% of those have adequate vessels in the upper arm. We knew that because if you have open flow in a loop graft, you have to have inflow and you have to have outflow. So the problem is it may not be in the location you wanted and you have to play a lot of games. But there is some blood flow draining it and some feeding it.
00:00

Replacing grafts with AV fistulas
And this kind of strategy has been used tremendously well out in the Spokane area by Vo Nguyen. And this is some of his data on creating secondary fistulas in patients with failing grafts. Look what he was able to do with his prevalence, up to 71%. This is a really nice strategy because you cannot hurt anybody. You already have the vessels. You have a very high probability of success.
00:00

Improvement targets
Now, let us go back and take all those things we talked about because I gave you a bunch of leverage points. Which ones are the most valuable? Which ones have the most bang for the buck? And where it is is in fistula creation and my friends are going to talk about that much more. You had high impact here doing monitoring with the gain 15 years ago. That was the sweet spot when we were at 2% catheters, 3% catheters. Now we have 30% catheters. So although this is very useful, you are going to have much higher impact if you work on fistula creation. Repair: The best repair is the one you do not need. And as long as we remember that we are going to have more impact here because you are will not need this; it does not mean you should not do this well. And again, secondary fistulas have very high impact.
00:00

Process dynamics
Many of you have seen this slide. I want to talk a little bit about process dynamics; what works. If you want to have something work, first you have to identify the risk group. So if we are talking about smoking cessation, define who smokes. It is really not useful to do a smoking cessation treatment program in people who do not smoke. So, you have to find who has the problem. It sounds stupid, but we do not always do that. Then you have to actually get them to go into their therapy. They have to enroll. And then the therapy or the intervention has some degree of efficacy. Our system, all the stuff I have talked about, is in here and only when you multiply across that do you get your impact.So, let us talk about fistula creation. I told you 80% of patients are candidates for fistulas. Only half of them ever get sent for an attempt. And if you do not map, only two-thirds of them ever work. If you multiply that across you get 27% fistulas; that is what you have. If you go around the country you have 27% fistulas and that is why.
Monitoring: If monitoring is 80% effective in terms of identification - and that actually flows a little better - and you actually send 80% of them over for an intervention, and if the intervention works 80% of the time, which is about what angioplasty does, you have a 50% impact. That is what you see in the programs. You drop the clotting rate 50%. That is what all the literature shows.
00:00

Shifting the burden
So how do we solve this? And I am going to open the floor and give it back to my friends who are going to talk about it. One of the things we have got to start talking about is what do solutions require? One of the things we have to deal with is shifting the burden strategy. The HMO that was in my town that had the open enrollment sign-up on the third floor of a walk-up worked very well.All right, so what we have to do is we have to align incentives. What you have to do is you have to incentivize the behavior you want. You have to have financial resources to cover added cost. It can be revenue neutral. We have shown over and over and over again that mapping pays for itself, monitoring pays for itself, but the savings are in the hospitals and you never get any of that money invested in the outpatient area. And you have to have options for places where the services are unavailable. There are towns all over the country where the surgeon says, "Look, I'm not doing it! It doesn't pay me. For $500 I don't want to come in at night. I would rather play bridge." Well, you have to have some resource to provide where there is a court of last resort and we have to talk about that.
00:00

Team approach
So what we are talking about is going from access process - creation, maintenance, repair and loss - to outcomes, to trying to do well for our patients. And this is the system we got adept to. And in each of these systems, there are all kinds of incentives and they do not necessarily align together and they align together at different times and in different ways. And so there is a probability distribution. You only have to have it not work for a small percentage of the patients for the cost to go through the roof. What we are trying to do is this: Work out a team approach. And the question becomes how do we do that? I am just going to go through that real briefly and then turn it over.
00:00

Potential improvements
Well, there are minor adjustments we can make. We can change the reimbursement a little bit; we can pay for preoperative imaging. Where I came from in Pennsylvania it was paid for but you had to do it within 24 hours. Well, that was great; you could not do it as an outpatient because you could never get it done within 24 hours. So what it was is there are always ways to write the regulation so that it looks good on paper but you cannot do it on the ground.Monitoring: There is no reason monitoring cannot be paid for, right? Medicare and CMS added it to the composite rate even though they had not even been thought about when the composite rate was sent.
Accountability at the source: What about a hospital or surgeon where 80% of their accesses are catheters? Is that okay? Why do we tolerate that? Why should that not be an accountability measure? Why should hospitals not be responsible for what they let out of the door? Why are they allowed to dump the problem downstream but if we have the problem what do we do? What is the easiest way to get above 40% fistulas in your unit? You take all the catheter patients, you put them in a bus and you send them to Chicago. Do you want the facility saying no to catheters? It is inappropriate. But if you set up a program in such a way you can incentivize facilities to say "I'm not taking that patient." That is not what this is about. What this is about is delivering services.
New measurements: We have to talk about what we measure. And do I really care if you have a fistula or a catheter? I only care if it affects outcome. And one of the problems of looking at things that are different than your outcome is, that sometimes the technology changes.
Where I am really interested is if I cannot provide enough blood flow to reach an adequacy target. That is a problem for me. I am interested if you do not show up for treatment. I am interested if you wind up in the hospital. I am interested if you die. If I have a catheter that never gets infected, that is fine as long as I can deliver adequate dialysis. Those are intermediate markers and we may need to start to think about this.
00:00

Financial metric = quality metric
Now, just on the side, when you talk about markers, you have to start to think about metrics in a different way. In my view, what I like to see is the purpose of operations is to drive quality. Because the way you set up your operations creates your quality and your quality outcome. And the purpose of your quality team is to streamline operations. And what brings that together is a metric and you want it to be a financial metric that is also a quality metric. I will give you an example and it is not related to access but it is an easy one. Reuse number.Reuse number in a reuse world, turns out to be a financial metric because if your reuse number goes down the financial people go bananas. And it is a quality metric. Well, how is it a quality metric? Well, if I wanted a dialyzer that does the job and can be reused, that is a problem. I have to have an inventory system, I have to have a process control in putting up the right dialyzer, I have to heparinize properly; you cannot get hypotensive or it clots and my reuse number goes down. I have to keep the people out of the hospital and I am setting up machines and I am losing dialyzers. I have to have a takeoff process control and I have to re-inspect that dialyzer so it keeps its surface area. So what happens is you have a financial metric that does exactly that. Operations drive quality and my quality team then streamlines all the interactions to drive operations. And it is tied up here. That is why it works. There are a lot of those numbers, but out of all the metrics, only some work and when you find that you can make an industry based on it.
00:00

Potential improvements
Last thing: Structural improvements. Regionalization; we talked about that. Somebody has to be responsible in a region. It may not be enough to have each facility try and handle it. Limited capitation, performing space measures, disease management, all of them discussed earlier this morning. We have to really talk about the system and deal with it on a larger scale than just dialysis facility because you are not going to fix it in a dialysis facility alone.
00:00

Conclusion
In conclusion, we have an opportunity to improve access care. We know what to do. We all know what to do. We can decrease the morbidity and mortality and decrease the cost but if we are going to have sustainable improvement we have to address the systemic factors that limit those improvements. And it can be best be accomplished by aligning incentives to reward and drive outcome improvements and we need to have everybody at the table. We have to go back to our roots and this has to be something that involves payers and facilities and physicians and patients, hospitals, everybody has to be at the table because unless we have everybody at the table it is hard to do.Thanks very much.
References
- Pisoni RL, Young EW, Dykstra DM, Greenwood RN, Hecking E, Gillespie B, Wolfe RA, Goodkin DA, Held PJ. Vascular access use in Europe and the United States: results from the DOPPS. Kidney Int 2002 Jan;61(1):305-16
- Dhingra RK, Young EW, Hulbert-Shearon TE, Leavey SF, Port FK. Type of vascular access and mortality in U.S. hemodialysis patients. Kidney Int 2001 Oct;60(4):1443-51
- Lok CE, Stanley KE, Hux JE, Richardson R, Tobe SW, Conly J. Hemodialysis infection prevention with polysporin ointment. J Am Soc Nephrol 2003 Jan;14(1):169-79
- Friedman AL, Walworth C, Meehan C, Wander H, Shemin D, DeSoi W, Kitsen J, Hill C, Lambert C, Mesler D. First hemodialysis access selection varies with patient acuity. Adv Ren Replace Ther 2000 Oct;7(4 Suppl 1):S4-10
- Silva MB Jr, Hobson RW 2nd, Pappas PJ, Jamil Z, Araki CT, Goldberg MC, Gwertzman G, Padberg FT Jr. A strategy for increasing use of autogenous hemodialysis access procedures: impact of preoperative noninvasive evaluation. J Vasc Surg 1998 Feb;27(2):302-7; discussion 307-8
- Gibson KD, Caps MT, Kohler TR, Hatsukami TS, Gillen DL, Aldassy M, Sherrard DJ, Stehman-Breen CO. Assessment of a policy to reduce placement of prosthetic hemodialysis access. Kidney Int 2001 Jun;59(6):2335-45
- McCarley P, Wingard RL, Shyr Y, Pettus W, Hakim RM, Ikizler TA. Vascular access blood flow monitoring reduces access morbidity and costs. Kidney Int 2001 Sep;60(3):1164-72
- McDougal G, Agarwal R. Clinical performance characteristics of hemodialysis graft monitoring. Kidney Int 2001 Aug;60(2):762-6
|
|||||||||||||