Kristensen SL, Docherty KF, Jhund P, et al.

Dapagliflozin Reduces the Risk of Hyperkalaemia in Patients with Heart Failure and Reduced Ejection Fraction: A Secondary Analysis from DAPA- HF

ASN Annual Meeting 2020 -- Digital Meeting
J Am Soc Nephrol (Oct) 31:21A 2020
BACKGROUND
Hyperkalaemia often limits the use of mineralocorticoid receptor antagonists (MRAs) in patients with heart failure and reduced ejection fraction (HFrEF), denying these patients a life-saving therapy.
METHODS
The risk of developing mild hyperkalaemia (potassium > 5.5 mmol/L) and moderate/severe hyperkalaemia (>6.0 mmol/L) was examined in the Dapagliflozin And Prevention of Adverse-outcomes in Heart Failure trial (DAPA-HF) according to background MRA use, and randomized treatment assignment, by use of Cox regression analyses.
RESULTS
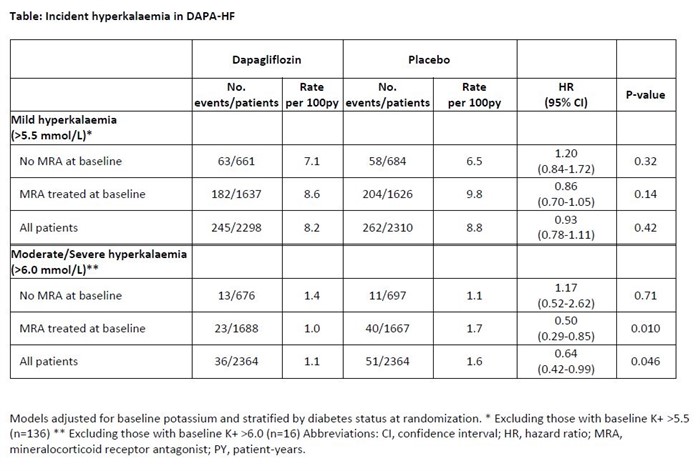
Overall, 3370 (70.1%) patients in DAPA-HF were treated with an MRA. Mild hyperkalaemia and moderate/severe hyperkalaemia occurred in 182 (11.1%) and 23 (1.4%) patients treated with dapagliflozin as compared to 204 (12.6%) and 40 (2.4%) of patients given placebo (Table and Figure). This yielded a hazard ratio (HR) of 0.86 (0.70- 1.05) for mild hyperkalaemia and 0.50 (0.29, 0.85) for moderate/severe hyperkalaemia, comparing dapagliflozin to placebo.
CONCLUSION
Patients with HFrEF and taking an MRA who were randomized to dapagliflozin had half the incidence of moderate/severe hyperkalaemia, compared with those randomized to placebo.
Incident hyperkalaemia in DAPA- HF
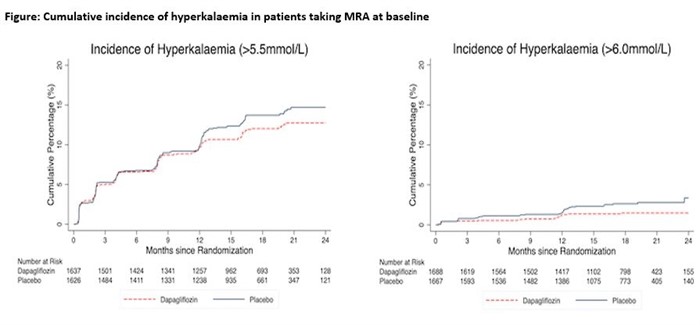
Cumulative incidence of hyperkalaemia in patients taking MRA at baseline
c Copyright 2020 -2021 American Society of Nephrology. Reproduced with permission.
All ASN abstracts from the 2020 Annual Meeting are available at this link and also are archived in .pdf form at ASN-Online.org
Disclaimer: Abstracts often have errors, both typographical and otherwise. This posting is an electronic translation of submitted abstracts which has not been verified against the original submitted abstract nor with the authors for accuracy. As a result, there may be errors, especially with regard to drug doses, but not limited to these. Abstracts undergo only limited review, and data often are changed as a result of the peer review process, so their reliability is less than manuscripts published in peer-reviewed journals. In using these summaries, you are agreeing that you are aware of these limitations.
The materials are provided on an as-is basis without any warranty of any kind, either express or implied. In addition to errors, the information presented may be incomplete or outdated. The information contained is not intended nor recommended as a substitute for professional medical advice. You are advised to check the appropriate medical literature and the product information currently provided by the manufacturer of each device to be used or drug to be administered to verify the dosage, the method and duration of administration, or contraindications. It is the responsibility of the treating physician or other health care professional, relying on independent experience and knowledge of the patient, to determine drug, disease, and the best treatment for the patient.
To the fullest extent permitted by law, HDCN, ASN and their affiliates and suppliers disclaim all warranties, express or implied, including, but not limited to, any warranty of merchantability, non- infringement or fitness for a particular purpose.
In no event shall HDCN, ASN, or their affiliates or suppliers be liable for any damages whatsoever (including, but not limited to, direct, indirect, incidental, consequential, punitive or exemplary damages, or any damages for loss of profits, use, data, goodwill or other intangibles) arising from or in any way relating to these terms, the materials, or any information, goods or services obtained from or referred to in the materials, whether based on warranty, contract, tort (including, but not limited to, negligence), or any other legal theory, and whether or not any or all of the limited entities is advised of the possibility of such damages.
Related Folders: