Rivara MB, Edwards TC, Patrick D, et al.

Development and Content Validity of a Patient-Reported Experience Measure for Home Dialysis

ASN Annual Meeting 2020 -- Digital Meeting
J Am Soc Nephrol (Oct) 31:51A 2020
BACKGROUND
The population of patients with kidney failure in the United States utilizing home dialysis modalities is growing rapidly. Unlike for in-center hemodialysis, there is no patient-reported experience measure for assessment of patient experience of care for peritoneal dialysis or home hemodialysis. We sought to develop and establish content validity of a patient-reported experience measure for patients undergoing home dialysis using a mixed-methods multiple stakeholder approach.
METHODS
We conducted a systematic literature review, followed by concept elicitation focus groups and interviews among 65 participants, including 21 home dialysis patients, 33 home dialysis nurses, 3 patient care partners, and 8 nephrologists. We generated a list of candidate items for possible measure inclusion, and conducted a national aspects of care prioritization exercise among 91 home dialysis patients and 39 providers using a web-based platform. We drafted the Home Dialysis Care Experience (Home-DCE) instrument and conducted 3 rounds of cognitive debriefing interviews to evaluate item comprehensibility, order, and structure. We iteratively refined the measure based on interview findings.
RESULTS
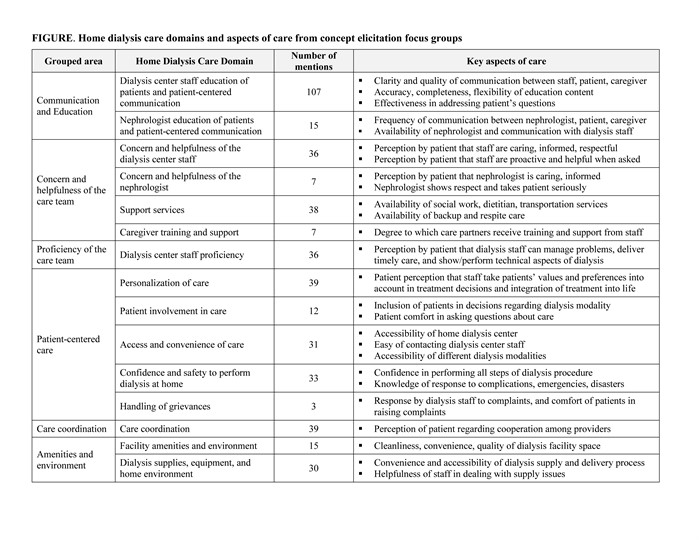
The literature review and concept elicitation phases supported 15 domains of home dialysis care experience in 6 general areas: communication and education of patients; concern and helpfulness of the care team; proficiency of the care team; patient-centered care; care coordination; and amenities and environment. Focus groups results showed that domains of highest importance for measure inclusion were home dialysis staff education and patient-centered communication, care coordination, and personalization of care (Figure). Aspects of care prioritization exercise results confirmed focus group findings. Cognitive debriefing indicated that the final measure was easily understood and supported content validity.
CONCLUSION
The Home- DCE instrument is a 26-item patient-reported experience measure for use in peritoneal dialysis and home hemodialysis. Qualitative focus group and prioritization survey data support measure content validity. To our knowledge, the Home-DCE instrument represents the first rigorously developed and content valid English language survey instrument for assessment of patient-reported experience of care in home dialysis.
c Copyright 2020 -2021 American Society of Nephrology. Reproduced with permission.
All ASN abstracts from the 2020 Annual Meeting are available at this link and also are archived in .pdf form at ASN-Online.org
Disclaimer: Abstracts often have errors, both typographical and otherwise. This posting is an electronic translation of submitted abstracts which has not been verified against the original submitted abstract nor with the authors for accuracy. As a result, there may be errors, especially with regard to drug doses, but not limited to these. Abstracts undergo only limited review, and data often are changed as a result of the peer review process, so their reliability is less than manuscripts published in peer-reviewed journals. In using these summaries, you are agreeing that you are aware of these limitations.
The materials are provided on an as-is basis without any warranty of any kind, either express or implied. In addition to errors, the information presented may be incomplete or outdated. The information contained is not intended nor recommended as a substitute for professional medical advice. You are advised to check the appropriate medical literature and the product information currently provided by the manufacturer of each device to be used or drug to be administered to verify the dosage, the method and duration of administration, or contraindications. It is the responsibility of the treating physician or other health care professional, relying on independent experience and knowledge of the patient, to determine drug, disease, and the best treatment for the patient.
To the fullest extent permitted by law, HDCN, ASN and their affiliates and suppliers disclaim all warranties, express or implied, including, but not limited to, any warranty of merchantability, non- infringement or fitness for a particular purpose.
In no event shall HDCN, ASN, or their affiliates or suppliers be liable for any damages whatsoever (including, but not limited to, direct, indirect, incidental, consequential, punitive or exemplary damages, or any damages for loss of profits, use, data, goodwill or other intangibles) arising from or in any way relating to these terms, the materials, or any information, goods or services obtained from or referred to in the materials, whether based on warranty, contract, tort (including, but not limited to, negligence), or any other legal theory, and whether or not any or all of the limited entities is advised of the possibility of such damages.
Related Folders: