Fakhouri F, Miller B, Scully M, et al.

Characteristics and Outcomes of Pregnancy-Triggered Atypical Hemolytic -Uremic Syndrome (aHUS): Global aHUS Registry Analysis

ASN Annual Meeting 2020 -- Digital Meeting
J Am Soc Nephrol (Oct) 31:55A 2020
BACKGROUND
Pregnancy-triggered aHUS (P-aHUS) accounts for 10–20% of aHUS diagnoses. Complement-mediated thrombotic microangiopathy (CM-TMA) may be associated with high maternal and fetal morbidity and mortality such as ESRD. The clinical characteristics of P-aHUS and survival probability in patients treated with the complement C5 inhibitor eculizumab are described, using the largest collection of P-aHUS data available in a single study.
METHODS
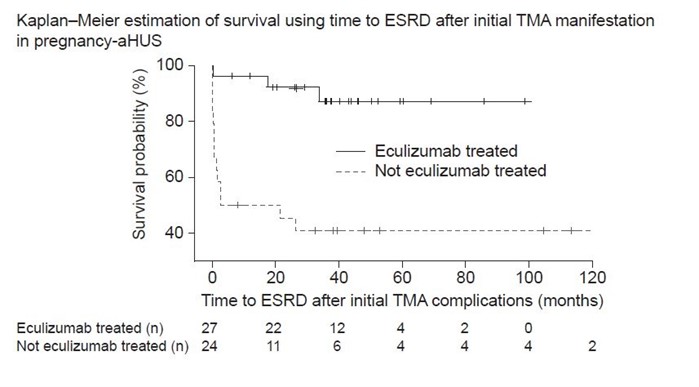
Patients with a clinical diagnosis of aHUS were included in the global aHUS registry (NCT01522183). Patients with P-aHUS were selected as those with first TMA manifestations during pregnancy or within 60 days postpartum. Patients with other triggers of aHUS were excluded. Survival, based on time to ESRD, was calculated by the Kaplan-Meier method.
RESULTS
In the registry, 51/1029 female patients were selected with P-aHUS and 27 received eculizumab. Mean ± SD age at pregnancy onset was 30.7 ± 5.9 years. P-aHUS occurred during pregnancy in 28 (54.9%) patients, with the remainder occurring postpartum. A diagnosis of pre-eclampsia or HELLP (hemolysis elevated liver enzymes low platelet count) syndrome was reported in 28 (54.9%) and 17 (33.3%) patients, respectively. A complement pathogenic variant was identified in 23 (45.1%) patients, of whom 3 (8.3%) also tested positive for anti-complement factor H antibodies. Mean ± SD eculizumab treatment duration was 1.8 ± 1.8 years. Survival probability was higher in eculizumab-treated patients compared with patients not receiving eculizumab (Figure).
CONCLUSION
Survival probability was higher in patients who received eculizumab compared with patients who did not receive eculizumab. Successful treatment with eculizumab, in addition to almost half of the patients having a complement pathogenic variant, confirms the appropriate classification of P-aHUS as a CM-TMA.
c Copyright 2020 -2021 American Society of Nephrology. Reproduced with permission.
All ASN abstracts from the 2020 Annual Meeting are available at this link and also are archived in .pdf form at ASN-Online.org
Disclaimer: Abstracts often have errors, both typographical and otherwise. This posting is an electronic translation of submitted abstracts which has not been verified against the original submitted abstract nor with the authors for accuracy. As a result, there may be errors, especially with regard to drug doses, but not limited to these. Abstracts undergo only limited review, and data often are changed as a result of the peer review process, so their reliability is less than manuscripts published in peer-reviewed journals. In using these summaries, you are agreeing that you are aware of these limitations.
The materials are provided on an as-is basis without any warranty of any kind, either express or implied. In addition to errors, the information presented may be incomplete or outdated. The information contained is not intended nor recommended as a substitute for professional medical advice. You are advised to check the appropriate medical literature and the product information currently provided by the manufacturer of each device to be used or drug to be administered to verify the dosage, the method and duration of administration, or contraindications. It is the responsibility of the treating physician or other health care professional, relying on independent experience and knowledge of the patient, to determine drug, disease, and the best treatment for the patient.
To the fullest extent permitted by law, HDCN, ASN and their affiliates and suppliers disclaim all warranties, express or implied, including, but not limited to, any warranty of merchantability, non- infringement or fitness for a particular purpose.
In no event shall HDCN, ASN, or their affiliates or suppliers be liable for any damages whatsoever (including, but not limited to, direct, indirect, incidental, consequential, punitive or exemplary damages, or any damages for loss of profits, use, data, goodwill or other intangibles) arising from or in any way relating to these terms, the materials, or any information, goods or services obtained from or referred to in the materials, whether based on warranty, contract, tort (including, but not limited to, negligence), or any other legal theory, and whether or not any or all of the limited entities is advised of the possibility of such damages.
Related Folders: