Pathologic features, nomenclature and diagnosis of small vessel vasculitis
Dr. Charles Jennette
May 14, 1997
 |
Charles Jennette, M.D.
Dr. Jennette is Professor of Pathology and Laboratory Medicine at the University of North Carolina at Chapel Hill. |

| Synchronized Real Media from Dr. Jennette's Presentation | ||

|
 28.8 modem click here to listen! |
This page is PART ONE. You can scroll through the talk while
listening to the audio. There are about 30 slides to this talk in
all, 15 in
part one.
Download this whole page before
starting up the audio player! The page will automatically
scroll to the slide being discussed. The hyperlink to PART TWO is
at the end of the PART
ONE.
You won't be able to listen to these files if you have not downloaded and configured the Real Video Player (it is built into Internet Explorer version 4.0). To download and configure the REAL player click here. |

Introduction of Dr. Jennette by Dr. Jörgen Wieslander:
You're all very welcome to this morning's session on systemic vasculitis. I'm very happy to introduce the first speaker, Professor Charles Jennette from Chapel Hill in North Carolina. Charles is the world's leading expert on the pathology of systemic vasculitis. In fact, he who together with Dr. Ron Falk formed the group that identified myeloperoxidase as one of the major antigens in the ANCA field.
Part One: Historical evolution of our concept of vasculitis
Dr. Charles Jennette
Thank you, Jörgen. I would like to thank the organizing committee for their invitation. It has certainly been a wonderful visit so far. And, of course, as a renal pathologist, it is really a distinct honor to be here in Scandinavia and to meet Klaus Brun, who obviously contributed to the origins of my subspecialty.
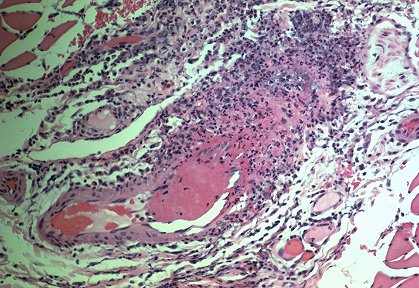
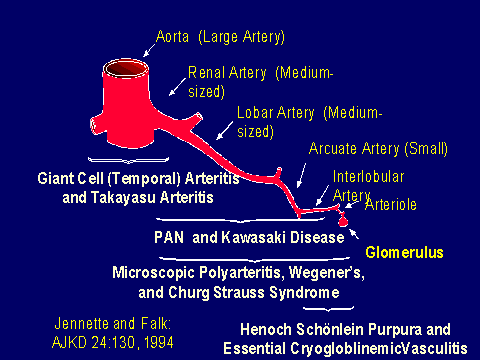
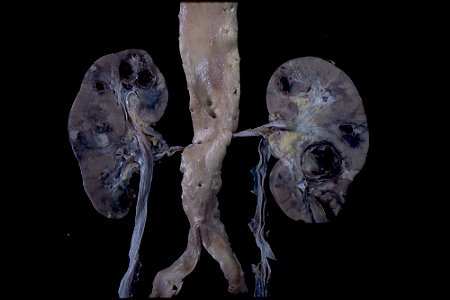
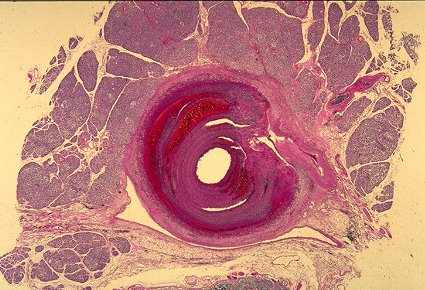
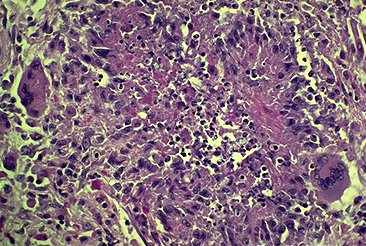
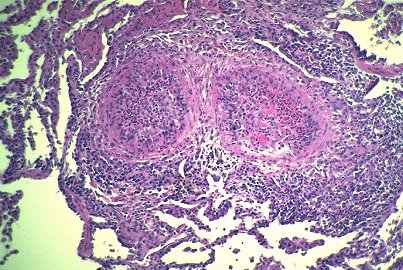
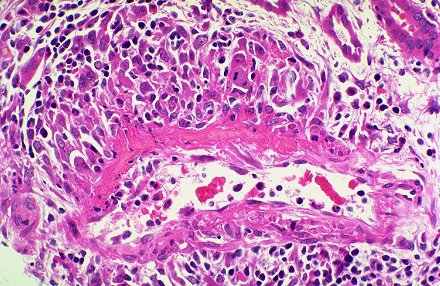
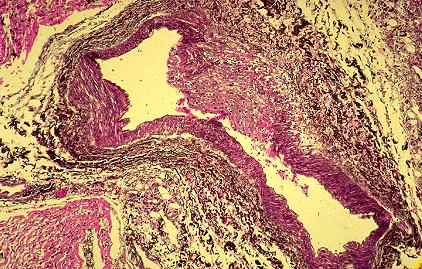
Vasculitis is characterized by inflammation in blood vessel walls
Systemic vasculitis, of course, as we are all aware, is a rather complex issue. Obviously in 30 minutes it is going to be difficult to really discuss this in great depth.